What is a medical or surgical mask?
A mask for medical or surgical use is a medical device. It traps droplets of saliva or airway secretions when the wearer exhales. It is for single use only. Worn by the caregiver : it protects the patient, the operating field or the equipment. Worn by the contagious person: it protects those around him/her.
What should be checked on the packaging of a medical or surgical mask?
A mask for medical or surgical use in Europe must indicate the reference to the standard EN 14683: 2019 + AC: 2019 with the type of mask claimed (see below).
What kind of performance requirements does this mask for medical or surgical use meet?
As indicated above, it is the EN 14683: 2019 + AC: 2019 standard that specifies the requirement levels. 4 performance criteria are expected to verify the quality of masks for medical or surgical use:
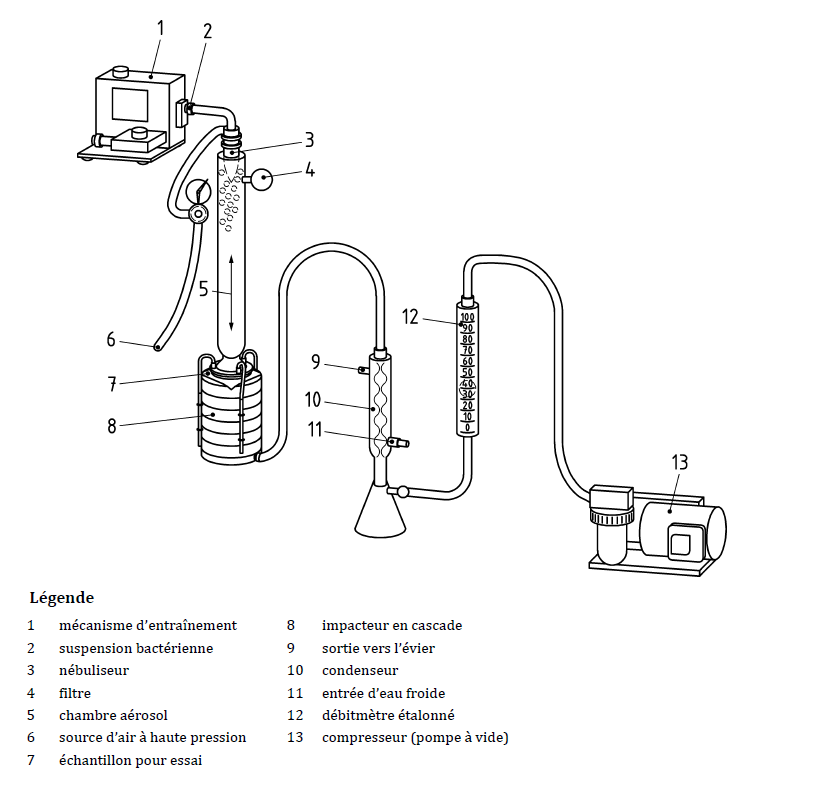
- Bacterial filtration efficiency (EFB or BFE)
- Determination of the breathability by measuring the differential pressure
- Microbial cleanliness
- Pressure of the splash resistance
Are there different performance levels between masks for medical and surgical use?
Yes, the EN 14683: 2019 + AC: 2019 standard distinguishes 3 types of masks:
Table 1 – Performance Requirements for Medical Masks
| Test | Type I* | Type II | Type IIR |
| Bacterial filtration efficiency (BFE), (%) | ≥ 95 | ≥ 98 | ≥ 98 |
| Differential pressure (Pa/cm²) | |||
| Splash resistance pressure (kPa) | Not required | Not required | ≥ 16,0 |
| Microbial cleanliness (cfu/g) | ≤ 30 | ≤ 30 | ≤ 30 |
Does HeX perform the tests according to EN 14683: 2019 + AC: 2019 ?
Yes, HeX performs its tests on masks for medical or surgical use according to the requirements of EN 14683: 2019 + AC: 2019
Table summarizing the tests proposed by HeX and compared to the requirements of EN 14683: 2019 + AC: 2019
| Test | Type I | Type II | Type IIR | Realized by HeX |
| Efficacité de filtration bactérienne (EFB ou BFE) | Required | Required | Required |  |
| Differential pressure | Required | Required | Required |  |
| Microbial cleanliness | Required | Required | Required |  |
| Pressure of the splash resistance | NO Required | NO Required | Required |  |
How does HeX ensure the quality of these test results?
Like most of the tests offered by HeX (more than 40) to its customers in the pharmaceutical and hospital industries, HeX performs these tests in accordance with the requirements of the ISO 17025 standard expected for testing, calibration and analysis laboratories.
Do I have to perform these tests in an accredited laboratory?
There is no regulatory or legal requirement in Europe for performance testing of masks for medical or surgical use to be performed by ISO 17025 accredited laboratories.
What are the trials proposed for the COVID-19 pandemic?
HeX offers to perform all 3 tests presented above, namely bacterial filtration efficiency, differential pressure and microbial cleanliness. In Belgium, a simplified protocol called ATP has been set up by the FAMHP (Federal Agency for Medicines and Health Products). Indeed, the FAMHP inspection services found that the proposed surgical mouth masks do not have the necessary declarations, certificates and test reports to demonstrate unequivocally that they meet the requirements of the applicable European standard (EN 14683: 2019 + AC: 2019) (extract from FAMHP website) This ATP protocol is thus based on the performance of one or two tests depending on the results. Only one laboratory is currently recognized as competent to perform these tests under the simplified ATP protocol.
What is the value of the test results from HeX compared to the results from Belgium’s ATP protocol?
The ATP protocol being by definition a simplified protocol compared to the EN 14683 : 2019 + AC : 2019 standard (number of tests and conditions of the tests degraded or simplified), the results resulting from test reports satisfying all the requirements of the EN 14683 : 2019 + AC : 2019 standard could not be opposed from a legal point of view